|
3/28/2024 0 Comments Non reactive gases periodic table 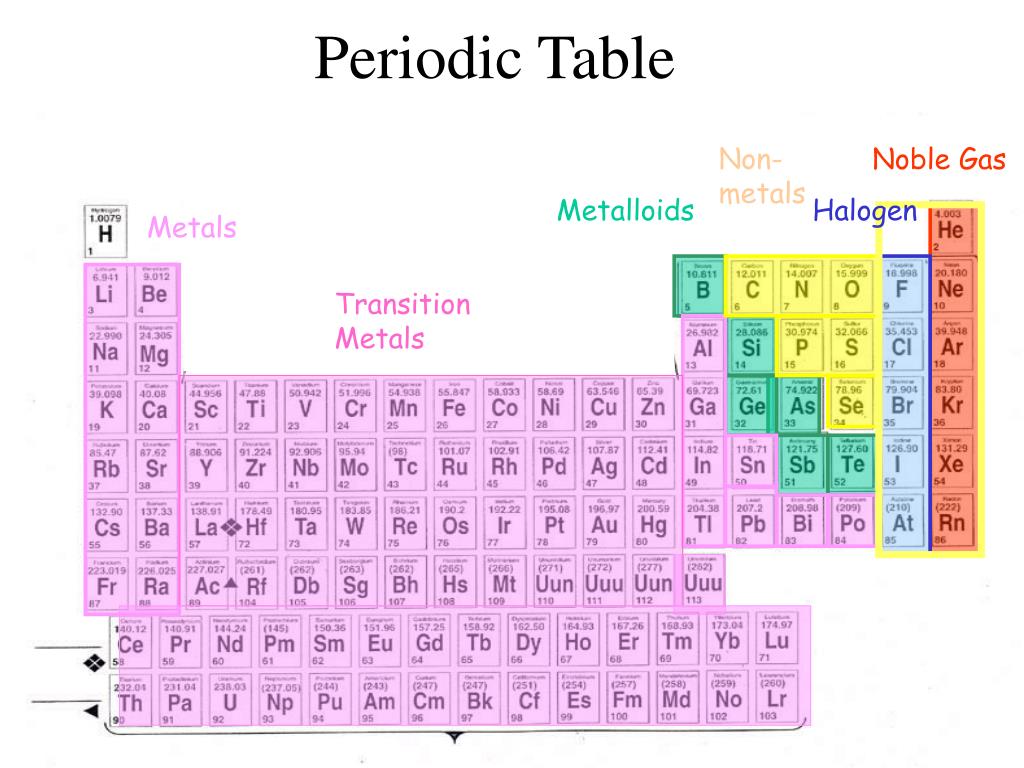
Inert Gases: Inert gasses are compounds that do not undergo chemical reactions. Difference Between Inert Gases and Noble Gases Definition Relationship Between Inert Gases and Noble GasesĪll noble gases are considered as inert gases under standard temperature and pressure conditions. In addition, Krypton can be chemically bonded to other nonmetals like hydrogen, carbon and with transition metals like copper. Krypton can also form fluorides like Xenon. Examples for Compounds Formed from Xenon: But, Argon, Krypton, Xenon are weakly reactive and can take part in compounds forming chemical bonds. Even under extreme conditions, Helium and Neon do not participate chemical bonding. All these gases are monoatomic gases under normal conditions. But under extreme conditions, they can be made into compounds. 
Other elements have s and p shells which are completely filled with 8 electrons. Thus, it has a maximum of 2 electrons in this orbital. This is because the atoms of these elements have completely filled valence shells. They show no or very low reactivity among other chemical elements. They are He (Helium), Ne (Neon), Ar (Argon), Kr (Krypton), Xe (Xenon) and Rn (Radon). Noble gases are the chemical elements in the group18 of the periodic table. Thus, there is no need to further react with other chemical species since all other atoms react with other chemical species in order to become stable by filling all the electron shells or by removing electrons in the outermost shell in order to obtain a complete valence shell. In other words, the outermost electron shells of the atoms of these gases are completely filled. The non-reactive behavior of inert gases arises due to the completed valence shells. It is a compound composed of two nitrogen atoms. Nitrogen is considered as an inert gas most of the times. Argon is a good example for an elemental inert gas. Inert gases can be elemental or can exist as compounds. Inert gasses are compounds that do not undergo chemical reactions. Key Terms: Inert Gas, Noble Gas, Hydrogen, Helium, Neon, Xenon, Nitrogen What are Inert Gases What is the Difference Between Inert Gases and Noble Gases What is the Relationship Between Inert Gases and Noble GasesĤ. The main difference between inert gases and noble gases is that inert gases do not undergo chemical reactions whereas noble gases can undergo chemical reactions at certain conditions. Gases are found as reactive gases and inert gases. Therefore, these particles are always in motion due to collisions that occur between particles. 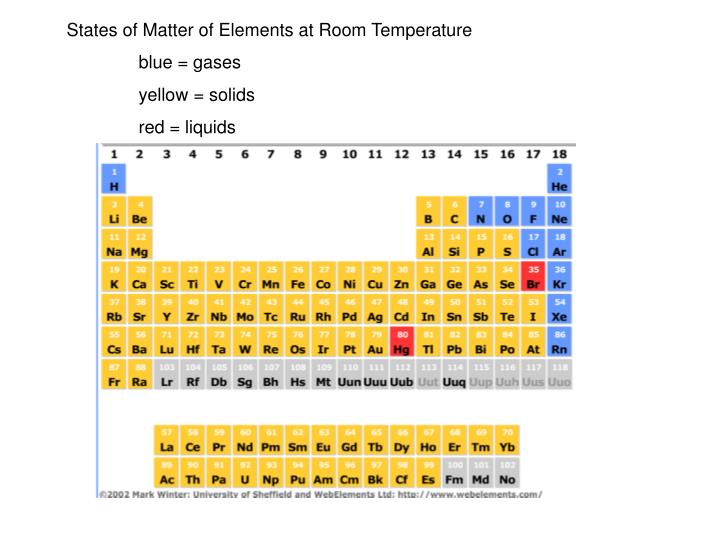
The attraction forces that exist between these gaseous particles are very less. However, gases are composed of tiny particles that have minute masses. Gaseous state includes gases that are either elements or compounds. Substances can exist in three major physical states known as the solid state, liquid state, and the gaseous state. H2S represents no considerable reactivity.Ī list of Reactive and Non-Reactive Gases to help when choosing Calibration or Bump test gas cylinders and accessories.Main Difference – Inert Gases vs Noble Gases Unlike non-reactive gases, reactive gases require higher flow (they will use a 1.0 LPM regulator, instead of a 0.5 LPM) and need corrosion-resistant equipment as corrosive and sticky (stainless steel regulators and Teflon tubing). The principal danger is that failure to use compatible materials and proper calibration procedures can result in dangerously inaccurate (low) readings and increased response times. Because of their greater tendency to be depleted from a gas sample by the exposed surfaces of gas detection systems, special care must be taken to ensure accurate monitoring results. Reactive gases are defined as gases which, because of their high chemical activity, are easily sorbed (adsorbed) by the exposed surfaces of gas detection systems including detector housings, calibration adapters, and remote sample draw accessories (tubing). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
